What Are Shed Exosomes in ARDS and Why Should You Care?
Understanding Acute Respiratory Distress Syndrome Basics
Acute Respiratory Distress Syndrome, or ARDS, is a sudden lung failure. It is a medical emergency. Healthy lungs move oxygen into your blood. They also remove carbon dioxide. ARDS damages this vital system. The lungs’ tiny air sacs, called alveoli, fill with fluid. This fluid comes from inflamed lung tissue. Imagine trying to breathe through a wet sponge. That is what ARDS feels like.
This condition does not happen on its own. It is a severe response to an injury or infection. Common triggers include: – A severe pneumonia infection. – Inhaling harmful substances like smoke. – A major infection spreading in the blood (sepsis). – A serious injury to the chest or other body parts.
The problem starts with widespread inflammation. The body’s defense system overreacts. It sends too many immune cells and signaling proteins to the lungs. This causes the walls of the blood vessels to leak. Fluid pours into the air spaces. Oxygen levels in the blood drop dangerously low. Patients cannot breathe on their own. They need help from a ventilator machine in an intensive care unit.
ARDS is serious and common. It affects hundreds of thousands of people each year. Even with modern care, about four out of every ten patients do not survive. Those who recover often face long-term health problems. They may have lasting shortness of breath and muscle weakness. Their brains may be affected, causing memory issues.
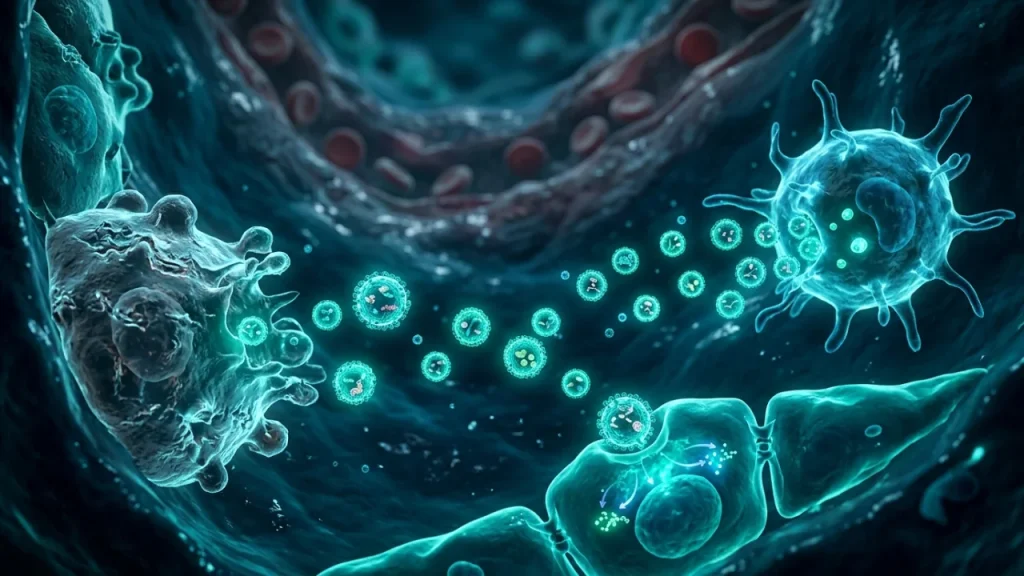
The key to better treatments lies in understanding the inflammation. Scientists now see it is not just about cells and proteins. Tiny messengers are crucial. Cells in the damaged lungs communicate by releasing vesicles. These are like microscopic bubbles carrying instructions. The most important type for signaling are called exosomes. During ARDS, cells shed exosomes at a high rate. These shed exosomes carry signals that can make inflammation worse. They can tell other cells to become more angry and destructive.
This is why ARDS research has shifted focus. The real damage comes from this cell-to-cell talk. By studying these messengers, scientists hope to find new ways to stop the cycle. They want to protect the lungs instead of just supporting them with a machine. Understanding this basic problem is the first step toward smarter treatments. The next section will explore these powerful messengers in detail.
Defining Exosomes: Nature’s Tiny Messengers
Imagine your body’s cells are like a vast city. They need to communicate quickly and precisely. They don’t use phones or emails. Instead, they send tiny physical packages. These packages are called exosomes.
Exosomes are incredibly small bubbles made by cells. They are about one-thousandth the width of a human hair. You could fit thousands of them on the period at the end of this sentence. Every cell in your body can make them. It is a normal, healthy process.
Cells create exosomes inside themselves. They pack these bubbles with important cargo. This cargo acts like instructions or news for other cells. The contents can include: – Proteins that act as signals or tools. – Genetic material like RNA, which carries blueprints. – Lipids and other molecules.
Once packed, the cell releases the exosome. It travels through bodily fluids like blood or lung fluid. Think of it like a bottle carrying a message in the ocean. The exosome floats until it finds another cell. That target cell accepts the package. It opens it and reads the instructions. Then it changes its behavior based on the message.
This system is vital for health. In a normal, healthy lung, exosomes help maintain balance. They carry signals for routine repair and calm communication between cells. They help the immune system stay alert but not overactive. It is a constant, quiet conversation that keeps tissues functioning well.
The term “shed exosomes” simply means exosomes that cells have released or let go. All exosomes are shed; it’s just the act of releasing them. In ARDS research, scientists pay close attention to these shed exosomes. They study what cargo they carry during sickness versus health.
The key point is this: exosomes themselves are not bad. They are neutral messengers. The problem arises when the cells sending them are damaged or inflamed. A stressed cell sends out different messages than a healthy one. It packs its exosomes with cargo that says “alert!” or “attack!” instead of “all is well.”
This changes everything in a disease like ARDS. The normal communication network gets hijacked. Helpful messages are replaced by harmful ones. The next section will show exactly how these corrupted messages drive the crisis in the lungs.
How Shed Exosomes Become Problematic in ARDS
In ARDS, inflamed lung cells release a flood of shed exosomes. These tiny vesicles are packed with dangerous cargo. They carry inflammatory signals, bits of damaged cell membrane, and specific microRNAs. These microRNAs are like master control switches for genes.
The target cells receive these harmful packages. The messages inside tell them to become angry and aggressive. This starts a vicious cycle that worsens lung injury. The process happens in several key steps.
First, these shed exosomes travel to immune cells called macrophages. In a healthy lung, macrophages clean up debris. The corrupted exosomes reprogram them. The macrophages become hyperactive. They release a storm of molecules called cytokines. This is a cytokine storm. It causes severe inflammation and fever.
Second, the exosomes target the endothelial cells. These cells line the blood vessels in the lungs. The harmful cargo makes these cells leaky. Fluid from the blood seeps into the tiny air sacs, the alveoli. This fluid is what causes pulmonary edema. It literally drowns the lungs from the inside.
Third, the exosomes damage the alveolar epithelial cells. These cells form the crucial barrier for air exchange. When they are injured, they cannot produce surfactant. Surfactant is a substance that keeps air sacs open. Without it, the sacs collapse. Breathing becomes incredibly difficult.
The cycle feeds itself. – Damaged cells shed more inflammatory exosomes. – These exosomes create more damaged cells. – Newly damaged cells then release even more harmful vesicles.
The normal repair signals are completely drowned out. The lung tissue becomes a battlefield. Scar tissue, called fibrosis, begins to form. This scarring makes the lungs stiff. Stiff lungs cannot expand properly to take in oxygen.
Research shows this exosome traffic is a main driver of ARDS severity. Scientists can measure the number of shed exosomes in a patient’s blood or lung fluid. Higher counts often mean worse outcomes. The cargo inside these vesicles acts as a fingerprint of the damage.
Understanding this process is crucial for one big reason. It gives us a new target for treatment. If we can intercept these harmful messages, we might stop the cycle. Therapies could block the release of bad exosomes. Other treatments could filter them from circulation. We could even engineer good exosomes to send repair signals.
This shifts the fight against ARDS. The goal is not just to support breathing with a ventilator. The new aim is to fix the broken cell conversation causing the crisis.
The Critical Link Between Exosomes and Lung Barrier Breakdown
The lung’s air sacs are protected by a delicate, living wall. This barrier has two main cell layers. The alveolar epithelial cells line the air side. The capillary endothelial cells line the blood side. They are held together by strong connections called tight junctions. This wall lets oxygen cross but keeps fluid out. In ARDS, this critical barrier fails. Fluid floods the air spaces. Shed exosomes are direct saboteurs of this wall.
These tiny vesicles carry molecular tools for damage. Their cargo includes active proteins and genetic signals. One major tool is a molecule called MMP-9. This is an enzyme that chews through the structural proteins of the cell wall. Think of it as molecular scissors. Exosomes deliver MMP-9 directly to the endothelial cells. The enzyme then cuts through the base that anchors the cells. The cell layer becomes leaky.
Simultaneously, exosome signals disrupt the tight junctions. They tell the cells to pull these connecting structures apart. The seals between cells break. Gaps form in the barrier. Plasma, the liquid part of blood, seeps through these new openings. It fills the precious air spaces where gas exchange must happen.
The damage does not stop at the blood vessel layer. Exosomes also attack the epithelial cells on the other side. These cells normally pump fluid out of the air spaces. Harmful exosome cargo can shut down these pumps. The injury becomes two-fold. Fluid pours in from the blood side. The system for removing it is disabled.
The process follows a clear sequence. – Inflammatory cells in the injured lung release shed exosomes. – These vesicles travel to the capillary endothelial cells. – They transfer MMP-9 and other disruptive signals. – The endothelial cell layer degrades and becomes porous. – Fluid leaks into the interstitial space and then into the alveoli. – Epithelial cell function is also impaired, preventing clearance.
This breakdown happens at a microscopic scale. But its effect is massive. The lungs, meant for air, fill with protein-rich fluid. This fluid also inactivates surfactant. Surfactant is the substance that keeps air sacs open. Its failure causes further collapse. Oxygen levels in the blood plummet.
Understanding this link is vital for care. It shows ARDS is not just widespread inflammation. It is a targeted assault on lung architecture. The exosomes are precise messengers of destruction. They convert a signal from an immune cell into physical rupture of tissue.
This knowledge guides new treatment paths. Researchers are testing molecules that block MMP-9. Other studies aim to protect tight junction proteins. The goal is to shield the barrier from the exosome attack. If we can fortify this wall, we could prevent the flooding that makes ARDS so severe. This mechanistic insight turns abstract inflammation into a solvable engineering problem within the body.
The Cellular Origins of Harmful Exosomes in ARDS
Epithelial Cells: First Responders Releasing Signals
The lung’s lining is not a passive victim. Its epithelial cells become active producers of harmful signals. These cells form the crucial barrier between air and blood. When viruses, bacteria, or toxins strike, they are the first to get injured. This injury triggers a frantic response. The cells do more than just die. They launch a coordinated communication effort. A key part of this effort involves shed exosomes.
Think of these cells as a besieged fortress. Their walls are under attack. Instead of just defending, they send out messengers. These messengers carry orders that can accidentally harm friendly troops. In ARDS, the epithelial cells release exosomes packed with specific cargo. This cargo includes: – Inflammatory molecules like cytokines. – Active enzymes that break down proteins. – Genetic instructions in the form of microRNAs.
This release happens very early in the injury process. It is a primary response, not a late effect. The exosomes travel to neighboring cells. They also enter the tiny blood vessels nearby. Their goal is to alert the immune system. However, the message often becomes distorted. The signal is too strong. It shifts from a call for help to a command for destruction.
The cargo inside these vesicles is particularly damaging. Enzymes like MMPs target the very structure holding cells together. They degrade the proteins that form tight junctions. These junctions are the seals between epithelial cells. Breaking them creates microscopic gaps. Fluid can then seep through these new openings.
Genetic material inside the exosomes alters cell function. MicroRNAs can switch off genes needed for repair. They can switch on genes that promote more inflammation. This reprograms receiving cells. A healthy neighbor cell might become inflammatory itself. It might even start producing its own harmful exosomes. This creates a dangerous cycle of signal amplification.
Research shows the quantity matters greatly. Injured epithelial cells can release ten times more exosomes than healthy cells. The number of vesicles correlates with the severity of the illness. More exosomes often mean worse barrier leak and lower oxygen levels. Scientists can measure these vesicles in fluid from patients’ lungs.
This process turns a local injury into a widespread problem. A few damaged cells can disrupt an entire lung region through these tiny packets. The exosomes act as force multipliers for damage. They ensure the initial insult has a long and destructive reach.
Understanding this origin is critical for new treatments. It suggests we could target the production or release of these vesicles right at the source. Stopping the signal at its start could prevent the cascade that follows. The epithelial cells are not just scenery in ARDS. They are central actors driving the disease forward through the exosomes they shed. This shifts our view from protecting a static barrier to calming an overactive signaling hub within it.
Immune Cells Amplifying the Inflammatory Storm
The initial alarm does not stay contained. Immune cells rushing to the injured lung become powerful secondary sources of harmful exosomes. These sentinel cells are designed to respond to threats. When they encounter signals from damaged lung tissue, they activate. This activation changes their entire mission. A key part of this new mission is to shed exosomes ards researchers now recognize as critical messengers.
Consider a macrophage, a large immune cell that acts as a cleaner. In a healthy lung, it quietly removes debris. In ARDS, it becomes inflamed. This activated macrophage starts packaging specific cargo into tiny vesicles. It does not release these exosomes at random. It targets them. Neighboring cells receive these molecular packages. The contents deliver precise instructions.
These immune-cell exosomes carry potent inflammatory signals. – They can contain proteins like cytokines trapped on their surface. – They often transport microRNAs that silence anti-inflammatory genes. – Some even carry pieces of bacterial membrane if an infection started the process.
This direct delivery is efficient and protected. Signals inside an exosome are not diluted in the fluid between cells. They are not easily broken down by enzymes. The vesicle acts as an armored truck. It ensures its dangerous cargo reaches the target cell intact. The receiving cell has no choice but to accept the delivery. It opens the package and follows the new orders.
The result is a rapid expansion of the inflammatory zone. One activated macrophage can influence dozens of other cells via exosomes. Those influenced cells may then signal others. This creates a network of communication that bypasses normal controls. The original injury site becomes a command center. It broadcasts orders through cellular packets.
Neutrophils, another key immune cell, contribute heavily to this storm. These cells are normally short-lived. In ARDS, they swarm the lungs and become persistently active. They also shed exosomes ards patients have in high numbers. Neutrophil-derived exosomes carry different tools. They contain enzymes that can directly damage lung matrix. They also send signals that attract even more neutrophils to the area. This creates a dangerous feedback loop.
The sheer volume becomes overwhelming. Immune cells can produce exosomes in massive quantities when fully activated. The lung fluid becomes crowded with billions of these vesicles. Each one is a potential spark for more inflammation. They turn the lung’s defense system against itself. The goal was to heal a local wound. The method instead causes widespread collateral damage.
This amplification stage explains why ARDS often worsens after the initial hit. The body’s own defense mechanisms overreact. They use exosomes to spread the alarm too far and too loudly. Stopping this cycle requires understanding both the source and the amplifiers. The next frontier is intercepting these messages before they rewrite cellular instructions across the entire organ.
Endothelial Cells and Vascular Damage Signals
The lungs are filled with tiny, delicate blood vessels. These vessels are lined with endothelial cells. In healthy lungs, these cells form a tight, selective barrier. Their job is to keep blood in and let gases pass through. In ARDS, this critical barrier fails. Endothelial cells become injured and activated. This activation changes their entire mission. They start to shed exosomes ards researchers now recognize as key damage signals.
These aren’t simple debris packets. They are loaded cargo. Injured endothelial cells pack their exosomes with specific instructions. The cargo tells other cells that the vascular system is in distress. One major signal is called von Willebrand Factor. This protein normally helps with clotting. When sent via exosome, it can trigger tiny clots in lung capillaries. Another common cargo is adhesion molecules. These molecules act like sticky flags.
The exosomes travel from the vessel lining into the blood and surrounding tissue. They deliver their sticky adhesion molecules to other cells. This makes immune cells like neutrophils stick to vessel walls more easily. It traps them right where they can cause the most harm. The exosomes also carry genetic instructions in the form of microRNAs.
These microRNAs can alter how recipient cells behave. For example, one type might tell a neighboring endothelial cell to become more permeable. This means the vessel wall gets leakier. Fluid from the blood seeps into the air sacs of the lung. This fluid buildup is a hallmark of ARDS. It directly causes low oxygen levels.
The process creates a vicious cycle locally. – Initial injury damages the endothelium. – Damaged cells release exosomes with pro-clotting and sticky signals. – These exosomes cause clotting and trap immune cells. – Trapped immune cells release more inflammatory signals. – This further injures more endothelial cells.
The cycle continues. The damage spreads along the vascular network. The exosomes act as both messenger and weapon. They transform a local leak into widespread system failure. The vascular barrier becomes like a shattered window. It can no longer keep the elements out.
This endothelial response links directly to the immune cell activity described earlier. Neutrophils stick to vessels primed by endothelial exosomes. Then they migrate into lung tissue and release their own damaging vesicles. Macrophages receive signals from endothelial exosomes too. They may become more inflammatory. The communication network now includes every major player in the lung.
Understanding this source is crucial for one reason. It points to a potential early intervention point. Vascular damage often starts the catastrophic chain reaction. If scientists could intercept these specific harmful exosomes, they might stabilize the barrier. A stable barrier could prevent much of the fluid flooding and inflammation that follows. The fight against ARDS requires silencing these false alarms from within the vessel walls themselves.
Comparing Exosome Sources and Their Unique Cargos
Not all exosomes in ARDS are the same. Their danger or benefit depends entirely on which cell created them. Different distressed cells pack different molecular instructions into these tiny vesicles. Think of them as specialized delivery trucks. Each truck type carries a unique cargo for a specific job.
Endothelial cells, lining blood vessels, shed exosomes loaded with clotting signals. These exosomes act like alarms. They tell platelets to form clots. They also tell immune cells to stick to the vessel wall. This starts the local leak described earlier.
Immune cells are another major source. Activated neutrophils release exosomes packed with destructive enzymes. – These enzymes can directly break down lung tissue. – They also carry signals that call more immune cells to the site. This creates a damaging feedback loop of inflammation.
Alveolar epithelial cells are different. These cells form the delicate air sacs of the lungs. When injured, they can release exosomes with healing signals. But they can also release vesicles with harmful proteins. These proteins may further disrupt the lung’s own repair systems.
Macrophages, the lung’s clean-up crew, add more complexity. They can release two broad exosome types. – Pro-inflammatory exosomes worsen tissue injury. – Anti-inflammatory exosomes may help calm the immune response. The balance between these types influences whether ARDS gets better or worse.
The cargo inside defines the exosome’s role. Harmful cargos often include specific molecules. One key molecule is Tissue Factor. This protein triggers the clotting cascade outside blood vessels. Another is specific microRNAs. These are tiny genetic switches. They can turn off genes needed for cell survival and barrier function.
Some exosomes carry “eat me” signals. These signals mark healthy cells for destruction by the immune system. Other vesicles carry molecules that make blood vessels more permeable. This directly leads to fluid leakage.
Scientists can now identify these different vesicles. They use the unique proteins on an exosome’s surface. These surface proteins act like a return address. They show which cell sent the vesicle.
This knowledge is vital for treatment. A therapy that blocks all exosomes might stop harmful messages. But it could also stop helpful ones. The goal is smarter targeting. Future treatments may only block exosomes from specific cells, like endothelial cells or overactive neutrophils.
Understanding these origins and cargos gives a clearer picture. It shows ARDS is not one conversation but many. Cells shout different orders into a crowded room. The result is chaos and collateral damage to the lungs. Silencing the most harmful voices in that crowd is the next frontier.
Molecular Cargo Inside ARDS Exosomes
Proteins That Trigger Inflammation Cascades
Exosomes carry powerful proteins that can start dangerous chain reactions. One major protein is called Tissue Factor. This protein is not usually active outside blood vessels. But exosomes can deliver it directly to lung tissue. There, it acts like a misplaced match. It ignites the clotting system where clotting should not happen.
This misplaced clotting creates tiny fiber webs. These webs are made of fibrin. They clog the delicate structures of the lungs. They also trap inflammatory cells. This creates a vicious cycle. More immune cells arrive. They get stuck and release more damaging signals. The lung’s ability to exchange oxygen gets worse.
Another key protein is High Mobility Group Box 1, or HMGB1. This is an alarm protein. Cells release it when they are severely stressed or dying. Exosomes can carry HMGB1 and protect it. This lets the alarm signal travel far from the original damage site.
When exosomes shed HMGB1 into new areas, immune cells react. They think the alarm is coming from there. This draws more inflammation to healthy tissue. It is like a false fire alarm pulling trucks to the wrong street.
Exosomes also carry direct signals for immune cells. One example is Interleukin-1β. This is a potent inflammatory message. An exosome can deliver it directly to another cell’s surface. This triggers that cell to release other inflammatory chemicals. The effect amplifies quickly.
- Tumor Necrosis Factor-alpha (TNF-α) is another signal.
- Transforming Growth Factor-beta (TGF-β) can push cells toward scarring.
- Specific enzymes called matrix metalloproteinases (MMPs) break down lung scaffolding.
These proteins do not work alone. They work in networks. One protein activates a cell. That cell then releases different exosomes with new proteins. The process feeds itself.
Scientists track these proteins by studying shed exosomes from patient fluid. Finding these proteins helps explain why inflammation spreads. It is not just free molecules floating around. The exosomes package them for delivery. This packaging makes the message stronger and longer-lasting.
Blocking these proteins is a treatment goal. Some experimental approaches try to catch the exosomes first. Others aim to neutralize the proteins after they are delivered. Knowing the exact cargo helps design better drugs.
The damage from these proteins is direct. They trigger clots, sound false alarms, and order more attacks. But exosomes also carry quieter, genetic instructions. These come in the form of microRNAs, which we will explore next.
Lipids and Their Role in Cell Membrane Disruption
Exosomes carry more than proteins and genetic codes. They also transport powerful lipid molecules. These fats are not just structural. They are active signals that can break down the lung’s vital barrier.
This barrier is made of tightly joined lung cells. It keeps fluid out of the air sacs. In ARDS, this seal fails. Fluid leaks in. Patients struggle to breathe. Shed exosomes from inflamed cells deliver lipids that cause this failure.
One major culprit is a lipid called sphingosine-1-phosphate (S1P). Its role is complex. At normal levels in the right place, S1P helps maintain barriers. But exosomes can deliver too much S1P to the wrong cells. This overload sends confused signals.
The signals disrupt the proteins that hold cells together. Think of these proteins as rivets and sealant. S1P tells the cell to remove these rivets. Gaps form between cells. Plasma and inflammation can then seep through.
Other lipids have direct toxic effects. Lysophosphatidic acid (LPA) is one example. This lipid can activate stress pathways inside lung cells. It tells cells to contract and pull away from neighbors. This further widens the gaps.
Ceramide is another key lipid. It is involved in creating the exosome itself. High ceramide levels also promote cell death. Exosomes rich in ceramide can deliver this death signal to healthy barrier cells.
The process is a cascade. An immune cell gets activated. It sheds exosomes packed with S1P and LPA. These vesicles travel to the lung lining. They fuse with a barrier cell or bind to its surface.
Their lipid cargo enters the cell’s signaling system. The cell’s internal skeleton rearranges. It changes shape. The tight junctions holding it to the next cell loosen. The barrier becomes porous.
Scientists find these damaging lipids in exosomes from patient fluid. Studying this cargo shows how barrier failure happens at a molecular level. It is not just brute force from inflammation. It is a precise chemical sabotage.
- Disrupted tight junctions let fluid pass.
- Changed cell shape creates larger gaps.
- Cell death signals thin the protective layer.
Blocking these lipid signals could plug the leak. Research looks at inhibitors for their receptors. The goal is to stop the signal before the cell obeys the disassembly order.
Lipids work with the proteins and microRNAs from other exosomes. Together, they dismantle the lung’s architecture from within. This multi-pronged attack makes ARDS so severe.
Understanding this cargo opens new treatment paths. Doctors could one day intercept these harmful vesicles. The focus shifts from just calming inflammation to directly protecting the barrier itself. This molecular insight is crucial for healing.
Nucleic Acids: microRNAs as Master Regulators
Exosomes carry more than just lipids and proteins. They also transport genetic instructions. These instructions come in the form of tiny molecules called microRNAs. Think of microRNAs as master switches for genes. A single microRNA can turn dozens of genes on or off.
During ARDS, immune cells shed exosomes packed with specific microRNAs. These vesicles deliver their cargo to lung barrier cells and other immune cells. The recipient cell then follows these new orders. Its normal function changes dramatically.
For example, one common microRNA found in ARDS exosomes is miR-155. This molecule is a powerful regulator of inflammation. When exosomes deliver miR-155 to a lung cell, it silences genes that calm the immune response. This allows inflammation to rage unchecked.
Another key player is miR-223. It often travels in exosomes from infected lungs. Its job is to reprogram how cells use energy. It pushes cells toward a state that produces more damaging reactive molecules. This worsens oxidative stress in already fragile tissue.
The process is a targeted takeover. – Exosomes from inflamed tissue seek specific cell types. – They fuse and release their microRNA cargo. – These microRNAs latch onto the cell’s own messenger RNAs. – This blocks the production of protective proteins. – The cell’s behavior shifts to promote damage and leakiness.
Scientists can find these microRNA signatures in patient blood or fluid. High levels of certain microRNAs link directly to worse outcomes. They are not just bystanders. They are active drivers of the disease.
Research shows blocking a single harmful microRNA can reduce injury in lab models. If you inhibit miR-155, for instance, inflammation decreases. The lung barrier stays stronger. This proves their master regulator role.
These tiny molecules work with the lipid signals we discussed earlier. Lipids like S1P change cell shape quickly. MicroRNAs change gene programs for the longer term. Together, they ensure the barrier breakdown is both rapid and sustained.
Understanding this offers huge hope for new treatments. Doctors could one day test for a “microRNA fingerprint” to gauge severity. Therapies might use artificial molecules to neutralize harmful microRNAs. The goal is to intercept the false instructions before cells obey them.
This turns exosomes from simple carriers into critical messengers of disease. Their nucleic acid cargo may hold the key to stopping ARDS at its genetic roots. Next, we see how proteins complete this destructive toolkit.
How Exosomal Cargo Reflects Lung Pathology
Exosomes shed within the inflamed lung carry a molecular snapshot of their parent cell’s condition. Think of them as tiny diagnostic packages. Their contents are not random. They directly reflect the stress and injury happening in the lung tissue.
During ARDS, different cell types become activated. Injured lung epithelial cells release one type of exosome. Angry immune cells like macrophages shed another. Each exosome population carries a distinct cargo signature. This cargo includes microRNAs, proteins, and lipids.
Scientists can analyze these shed exosomes from patient fluid samples. The cargo profile tells a detailed story. It reveals which cells are most active in the damage process. More importantly, it shows the severity of the pathology.
For example, exosomes from severe ARDS patients show clear cargo changes. – Levels of pro-inflammatory signaling proteins are often very high. – MicroRNAs that promote cell death are more abundant. – Protective repair molecules are frequently missing. This cargo mix directly mirrors the destructive activity in the lungs.
The quantity of exosomes also matters. More severe tissue injury usually means cells shed more exosomes. The body’s communication network goes into overdrive. Researchers see a correlation between exosome concentration in blood and clinical scores of lung dysfunction.
Specific cargo molecules serve as biomarkers. High levels of a protein like RAGE in exosomes can signal severe epithelial damage. An increase in certain microRNAs correlates with prolonged ventilator need. These are not just associations. The exosomal cargo is a functional part of the disease process.
This makes exosomes powerful diagnostic tools. A blood test could analyze shed exosomes to gauge real-time lung status. It could be faster and more precise than some current methods. Doctors could see which harmful pathways are most active in a specific patient.
The cargo profile also helps predict outcomes. A patient whose exosomes carry strong repair signals might have a better prognosis. Another patient with a cargo full of inflammatory molecules might need more aggressive treatment. This is personalized medicine at a molecular level.
Understanding this reflection is key for therapy. If exosomes mirror pathology, then changing their cargo could alter the disease course. Future treatments might aim to modify what cells pack into these vesicles. The goal is to shift the message from “cause damage” to “promote repair.”
In essence, the exosome is a messenger and a mirror. Its contents provide a window into the cellular battlefield of ARDS. This insight bridges our understanding to the next logical step: targeting these communication vesicles to intercept their harmful instructions.
Mechanisms of Exosome-Mediated Damage in ARDS
Propagating Inflammatory Signals Across the Lung
In ARDS, inflammation does not stay in one place. It spreads. Cells in a damaged lung area release millions of shed exosomes loaded with inflammatory molecules. These tiny vesicles travel through fluid in the lung’s air sacs. They also enter the bloodstream. Their cargo acts like a set of precise instructions. These instructions are delivered directly to other cells. This turns local damage into a widespread problem.
Think of it like a wildfire. A single spark starts the blaze. Exosomes are the embers carried by the wind. They land on fresh fuel and start new fires far from the original burn. In the lung, the “fuel” is healthy epithelial and endothelial cells. The “embers” are exosomes containing specific signals.
What is inside these vesicles? The cargo is designed to cause trouble. – Pro-inflammatory cytokines like IL-1β and TNF-α. These molecules shout “alert!” to immune cells. – Active fragments of damaged cell membranes that further trigger immune responses. – Specific microRNAs that silence genes for calming inflammation.
A healthy cell receives this exosome. It fuses with the vesicle and unloads its cargo. The cell’s normal functions are hijacked. It now follows new orders. The cell itself starts producing the same inflammatory signals. Then it packages those signals into its own exosomes. It sends them out again. This creates a vicious cycle. One signaling event becomes ten. Ten become a hundred.
The result is a coordinated attack on the lung’s architecture. Two main processes escalate. First, the blood vessels become leaky. Inflammatory exosomes target endothelial cells lining these vessels. Signals break down the tight seals between cells. Fluid from the blood pours into the air spaces. This is pulmonary edema. It floods the lungs and causes respiratory failure. Second, immune cells swarm the area. Exosomes call in neutrophils and macrophages. These cells aim to fight damage but often cause more harm. They release toxic chemicals and more exosomes.
This propagation explains why ARDS can worsen rapidly. The initial injury might be limited. Exosome communication systems the damage across both lungs. They ensure that inflammation becomes self-sustaining. The body’s own messaging system is turned against it.
Therefore, stopping this chain reaction is a key treatment goal. Scientists are looking for ways to block exosome release or uptake. Another strategy is to neutralize their harmful cargo before it delivers its message. Breaking this communication loop could slow the inflammatory fire. It could give the lung a chance to begin repair. This direct interference leads us to the next critical question: how can we design therapies to capture or reprogram these rogue messengers?
Direct Attack on the Alveolar-Capillary Barrier
The alveolar-capillary barrier is a fragile, essential wall. It is only two cells thick. On one side, tiny air sacs called alveoli hold oxygen. On the other side, capillaries carry blood. This thin wall lets oxygen move into the blood. It also keeps fluid in the vessels. In ARDS, this barrier fails catastrophically. Exosomes shed from inflamed cells are central to this failure. They execute a direct attack on both cell layers.
The assault begins on the capillary side. Endothelial cells form the blood vessel lining. Inflammatory exosomes bind to these cells. They deliver signals that disrupt the cell’s internal skeleton. This skeleton, called the cytoskeleton, gives the cell its shape and strength. When it contracts or falls apart, the cell shrinks. Gaps open between neighboring endothelial cells. These gaps are microscopic holes in the vessel wall.
Plasma, the liquid part of blood, now leaks through these gaps. It floods into the interstitial space between the vessels and air sacs. This fluid contains proteins like fibrinogen. The exosome signals also make the endothelial surface sticky. This stickiness traps platelets and forms micro-clots. Blood flow to healthy lung areas gets blocked.
The attack then crosses to the air sac side. Epithelial cells form the lining of the alveoli. These cells have two critical jobs. They facilitate gas exchange. They also actively pump fluid out of the air spaces to keep them dry. Harmful exosomes reach these epithelial cells. The exosome cargo damages the cell’s pumping machinery. Sodium channels and pumps stop working. The lung cannot remove the incoming flood.
Simultaneously, the exosome signals damage tight junctions. These are protein seals that stitch epithelial cells together. With broken seals and no pumping, fluid has a clear path. The plasma from the capillaries surges into the alveoli itself. The air sacs, meant for gas, fill with liquid.
This fluid has devastating effects: – It physically blocks oxygen from reaching the capillary blood. – It washes away surfactant, a substance that keeps alveoli open. – The protein-rich fluid becomes a breeding ground for more inflammation. – Stiff, heavy lungs require extreme ventilator pressure to inflate.
The result is the hallmark of ARDS: stiff, heavy, non-functional lungs. The patient hypoxemia deepens despite high oxygen support. The body’s own shed exosomes turned a cellular communication system into a wrecking crew. They did not just spread an inflammatory message. They physically dismantled the lung’s core structure cell by cell.
Therefore, therapeutic strategies must look beyond calming inflammation. They must aim to protect or rapidly repair this specific barrier. Stopping exosomes could plug the leaks at their source.
Fibrotic Remodeling: When Healing Goes Wrong
The initial crisis of flooding is just the first act. The body then tries to heal the injured lung. This repair process often goes badly wrong. Shed exosomes are central to this failure. They push the lung toward making too much scar tissue.
This scar tissue is called fibrosis. It is stiff and thick. It replaces the delicate, elastic walls of the alveoli. Think of a soft sponge turning into a tough piece of leather. A fibrotic lung cannot expand properly. It loses its ability to transfer oxygen forever.
Exosomes direct this harmful remodeling. They carry specific instructions to cells called fibroblasts. Fibroblasts are the body’s construction workers for tissue repair. Normally, they make a temporary scaffold of collagen to help healing. Then they stop. Exosome signals make them overwork.
The messages inside these vesicles tell fibroblasts to activate and multiply. Key signals include growth factors like TGF-β and specific microRNAs. These signals are packed into exosomes at the injury site. Inflamed lung cells release them.
The exosomes travel to resident fibroblasts. The fibroblasts receive the “build” command. They start producing massive amounts of collagen and other fibrous proteins. They do not get the signal to stop. The construction job never ends.
The result is a dense, tangled web of scar tissue. This fibrosis has several concrete effects: – It thickens the walls between air sacs, creating a longer path for oxygen. – It makes lung tissue stiff, increasing the work of breathing. – It permanently reduces the total space available for gas exchange. – It destroys the intricate capillary network around alveoli.
This process creates a vicious cycle. More scarring leads to more stiffness. Stiffness causes further injury from mechanical ventilation. This new injury prompts more exosome release. The cycle of fibrosis continues.
The origin of these fibrotic exosomes is critical. They come from many cells involved in the prolonged inflammation. Key sources include: – Activated immune cells like macrophages. – Injured alveolar epithelial cells. – Damaged endothelial cells from capillaries.
Each source adds its own damaging cargo to the exosomes. Together, they overwhelm the normal healing pathways. The goal of repair shifts from restoration to rigid scarring.
Stopping this fibrotic remodeling is a major therapeutic target. Researchers ask a key question. Can we intercept these harmful exosomes? Could we block their signals to fibroblasts? The aim is to guide healing back toward proper regeneration instead of scarring.
This fibrotic stage explains why many ARDS survivors face long-term disability. Their lungs are left permanently damaged. The battle is not just about surviving the initial storm. It is about preventing the frozen landscape of scar tissue that follows. Understanding exosome-driven fibrosis is the first step to stopping it.
The Vicious Cycle of Exosome Release and Tissue Injury
Injury to a single lung cell starts a dangerous chain reaction. This damaged cell does not suffer quietly. It sends out urgent signals. These signals are packaged inside tiny vesicles called exosomes. Think of them as microscopic distress packets.
The cell sheds exosomes loaded with specific cargo. This cargo includes: – Inflammatory proteins called cytokines. – Signals that trigger cell death. – Molecules that break down tissue.
These exosomes travel through the fluid between cells. They seek out other healthy cells nearby. The exosomes then fuse with the membranes of these healthy cells. They deliver their harmful cargo directly inside.
The result is predictable. The once-healthy neighbor cell now becomes stressed. It gets damaged by the inflammatory signals. This new damage forces it to send its own exosomes. It begins to shed exosomes too.
This creates a self-amplifying loop. One injured cell can corrupt several others. Those newly damaged cells then corrupt more. The wave of injury spreads across the lung tissue. It moves far from the original site of harm.
The process has clear steps. First, an insult like infection or trauma hurts alveolar cells. Second, these cells release exosomes with damage signals. Third, the exosomes bind to and activate immune cells like macrophages. Fourth, these activated macrophages release more potent exosomes. Fifth, the cycle repeats and expands.
The speed of this cycle is critical. More exosomes mean faster spreading damage. The lung’s delicate architecture is attacked from within. The communication system meant for repair is hijacked. It becomes a tool for spreading chaos.
Research shows this cycle is a key reason ARDS escalates so quickly. Blocking exosome release in lab models slows the injury. It limits the spread of inflammation. This finding highlights the cycle’s importance.
The content of these early exosomes is different from later fibrotic ones. Early on, the signals scream “inflame!” Later, they whisper “scar.” But the mechanism of spread remains the same. The relentless shedding drives the disease forward.
Therefore, the initial injury is only the first event. The sustained damage comes from this continuous communication. Exosomes are the messengers carrying bad news from cell to cell. Interrupting this conversation could stop the avalanche before it gains power. Understanding this cycle points to a clear strategy: break the chain of communication to protect the lung.
Diagnostic Potential of Exosomes in ARDS
Exosomes as Early Warning Biomarkers
Doctors need better tools to spot ARDS early. Current methods often rely on seeing clear lung damage on scans. By then, the harmful cycle is already active. Researchers now see shed exosomes as a potential solution. These tiny messengers travel in bodily fluids like blood. They carry molecular cargo from their parent lung cells. This cargo reflects the cell’s real-time state.
Think of exosomes as urgent text messages sent from the lung. We can intercept these messages. Analyzing them gives a direct report from the battlefield inside the alveoli. The content changes as the disease starts. This change happens before standard tests can confirm ARDS.
What specific signals do these exosomes carry? Their cargo includes distinct molecules. – Unique microRNAs that control inflammation. – Damaged proteins from stressed lung cells. – Surface markers identifying which cell type shed them.
A healthy person’s blood has a baseline level of exosomes. In early ARDS, the number of certain exosomes spikes rapidly. Their molecular cargo also shifts dramatically. This provides two key pieces of evidence: quantity and content.
The diagnostic potential is powerful. A blood test could detect these warning signs hours or days sooner. Early detection changes everything. Treatment could start before widespread lung injury occurs. It could prevent the worst damage.
This approach is like checking smoke before seeing flames. The exosomes are the smoke. Traditional scans only see the fire. Finding these biomarkers is a major research focus. Several candidate signals show high promise in studies.
The technical process involves collecting a small blood sample. Exosomes are separated and concentrated from the plasma. Scientists then analyze their contents using precise tools. They look for the signature of early alveolar distress.
Challenges remain, of course. Doctors must distinguish ARDS exosomes from those related to other conditions. A simple infection might also increase exosome counts. The true diagnostic test must find the unique ARDS signature. Work is ongoing to define this exact fingerprint.
The goal is a fast, accurate test for the ICU. It would help doctors make quicker, more confident decisions. This could directly improve patient outcomes. Catching ARDS at its start means therapies have a better chance to work.
Therefore, the same vesicles that spread injury also hold the key to early warning. Intercepting these messages offers a window into the lung’s hidden state. This turns a mechanism of disease into a tool for healing. The next step is using this knowledge not just to diagnose, but to guide precise treatment.
Analyzing Exosomal Signatures in Blood and Fluid
Doctors search for these critical exosomes in two main places: blood and lung fluid. Blood is the easiest and most common source. A standard blood draw provides plasma. Plasma is the liquid part of blood after removing cells. This liquid is rich in exosomes from all over the body. Finding the ARDS-specific ones is the key challenge.
Lung fluid offers a more direct signal. In severe cases, doctors may use a procedure called bronchoalveolar lavage. A thin tube is passed into the airways. A small amount of sterile fluid is washed into the lung and then collected. This fluid comes directly from the air sacs. Exosomes found here are likely from the injured lung tissue itself. They provide a clearer local picture.
The analysis focuses on what these shed exosomes carry. Each vesicle has a cargo and a surface. Scientists look at both.
First, they examine surface proteins. These are like mailing labels. They show which cell sent the exosome. An exosome from a damaged lung cell has different labels than one from a liver cell. Identifying these labels confirms the origin.
Second, they analyze the internal cargo. This cargo can include: – MicroRNAs, which are tiny genetic instructions that can turn other cell functions on or off. – Inflammatory proteins called cytokines that fuel the immune response. – Bits of damaged cell membranes that signal severe injury.
The pattern of these molecules creates a signature. This signature is the biomarker. A healthy person’s exosomes have one pattern. A patient developing ARDS has a different one. The diagnostic goal is to spot the ARDS pattern early.
The process requires several steps after sample collection. The exosomes must be isolated from the complex fluid. They are incredibly small. Special machines use high-speed spinning to concentrate them. Then, even more precise tools break them open to study their contents.
Researchers compare samples from many patients. They look for common signals in those who progress to severe ARDS. These repeated signals become the target for a future test.
Using both blood and lung fluid gives a fuller story. Blood tests are simpler for frequent monitoring. Lung fluid analysis gives deeper, localized data. Together, they can confirm a diagnosis.
This work turns vague suspicion into specific evidence. Instead of waiting for full-blown lung failure on a scan, doctors could see its molecular shadow first. They find these shed exosomes in ARDS research as vital clues. The next frontier is not just finding these signals but understanding exactly what they are telling cells to do. This knowledge could lead to treatments that block the harmful messages themselves.
Distinguishing ARDS from Other Lung Conditions
One major hurdle in the ICU is telling ARDS apart from other serious lung problems. Conditions like severe pneumonia or heart failure can look very similar on a chest X-ray. Patients with these issues may also struggle to breathe. This makes an accurate, swift diagnosis difficult. Traditional tests often cannot provide a clear answer quickly enough.
This is where exosome analysis shows strong promise. The cargo inside these tiny vesicles acts like a detailed molecular fingerprint. Different diseases create different fingerprints. The exosomes shed in ARDS carry a specific mix of signals. This mix reflects the unique inflammatory storm and lung tissue damage of ARDS.
Think of it like listening to an orchestra. Pneumonia might feature a loud section of bacterial alarm signals. Heart failure could highlight markers of fluid pressure stress. ARDS has its own distinct symphony of damage. The exosomes broadcast this symphony into bodily fluids.
Researchers are now cataloging these differences. They compare exosomes from patients with confirmed ARDS to those with other conditions. Early studies show distinct patterns in the microRNA and protein content. For instance, the balance of certain inflammatory proteins inside exosomes may differ.
- Exosomes from ARDS patients might carry more markers linked to widespread endothelial cell damage.
- Exosomes from bacterial pneumonia may contain stronger signals related to specific pathogen defenses.
- Exosomes in heart failure could be richer in indicators of mechanical strain.
This specificity is the key to a better diagnostic tool. A test based on these shed exosomes would not just detect lung inflammation. It would classify the type of inflammation. It could point directly to ARDS.
Such precision changes clinical decisions. Doctors could start targeted ARDS therapies faster. They could also avoid giving those potent treatments to patients who do not have ARDS. This prevents unnecessary side effects.
The goal is a definitive biological signature. This signature would move diagnosis beyond vague imaging and clinical scores. It offers a clear window into the cellular processes at play. This molecular clarity is what makes exosome research so compelling for ARDS diagnosis.
Ultimately, distinguishing these conditions early saves lives and resources. It directs the right care to the right patient at the right time. The next step is validating these signature patterns in larger patient groups to build a reliable test.
Monitoring Disease Progression Through Exosome Changes
Exosomes provide a moving picture of a patient’s condition. Their numbers and cargo change as the disease evolves. This makes them powerful tools for monitoring. Doctors can track treatment response in real time.
Think of it like a blood test. A standard test gives a single snapshot. But repeated exosome analysis creates a film. It shows the cellular conversation over days or hours. This is vital in a fast-changing illness like ARDS.
A patient might start with a flood of inflammatory shed exosomes. These vesicles carry signals that worsen lung injury. Effective treatment should slow this flood. The exosome count in the blood should drop. Their harmful cargo should decrease too.
Scientists look for specific changes in exosome content. These changes signal healing or decline.
- A drop in microRNAs that promote cell death is a good sign.
- An increase in proteins that help repair tissue shows progress.
- A shift in surface markers points to calmer blood vessel walls.
Monitoring these shifts offers objective data. It moves beyond just watching ventilator settings. It shows if the body’s biology is truly improving.
For example, a doctor uses a strong anti-inflammatory drug. Current methods take time to show effect. But exosome analysis might reveal a quicker molecular response. The vesicles could show reduced inflammatory signals within hours. This confirms the treatment is working at the cellular level.
Conversely, a rise in damaging exosomes warns of trouble. It can happen before traditional signs like falling oxygen levels. This gives doctors an early alert. They can adjust therapy before the patient gets much worse.
This approach personalizes critical care. Not all patients respond the same way. Two people with ARDS might need different treatments. Tracking their unique exosome profiles guides this choice. It helps find the right therapy for each individual.
The process is repeatable and minimally invasive. Exosomes can be collected from simple blood draws. Regular sampling builds a detailed timeline of the disease. This timeline is invaluable for making smart decisions in the ICU.
Ultimately, this turns exosomes into a treatment compass. They point the way toward recovery or signal a dangerous turn. This dynamic tracking could shorten ICU stays. It helps stop ineffective treatments faster. It directs resources to where they are most needed.
The next challenge is to standardize these monitoring protocols for widespread hospital use.
Therapeutic Strategies Targeting Exosomes in ARDS
Blocking Harmful Exosome Production and Release
Scientists are exploring ways to stop harmful exosomes at their source. This means preventing sick cells from making and releasing these damaging vesicles. The goal is to break a cycle of inflammation in the lungs.
One strategy targets the machinery inside the cell. Cells use special proteins to package exosomes. Researchers are designing drugs that block these proteins. Without them, the cell cannot form the bad exosomes. It is like taking a key part out of an assembly line.
Another approach stops the release. Cells must move exosomes to their outer membrane to shed them. Certain chemical signals control this process. Drugs can interfere with these signals. This traps the harmful exosomes inside the cell. They cannot escape to cause damage.
The body’s own cells sometimes send the wrong messages. In ARDS, stressed lung cells tell immune cells to attack. They do this by sending specific exosomes. Blocking these exosomes can calm the immune response. It tells the immune system to stand down.
Some experimental methods are very direct. They use tiny particles designed to bind to bad exosomes. These particles act like decoys. They capture the exosomes before they reach other cells. This neutralizes the threat in the bloodstream.
Key steps in a blocking strategy might include: – Identifying the specific harmful signals inside the exosomes. – Developing a drug that stops the cell’s packaging factory. – Testing if blocking release reduces lung injury in models. – Ensuring healthy cell communication is not harmed.
This work focuses on precision. The aim is not to stop all exosome production. That would be bad because exosomes have important jobs. The goal is to stop only the dangerous ones. This requires understanding their unique signatures.
The timing of treatment is also critical. Blocking might work best early in the disease. It could prevent the inflammation from spiraling out of control. Later, different strategies might be needed.
Challenges remain. Cells might find other ways to send signals. Therapies must be tested for safety over time. But the principle is clear: intercepting bad messages can protect the lungs.
Successfully blocking harmful exosome production and release could change ARDS care. It moves us from managing damage to preventing it. This approach tackles a root cause of the illness. The next logical step is to ask what to do with the harmful exosomes already circulating in the body.
Engineering Protective Exosomes for Treatment
Scientists can now design helpful exosomes in the lab. These are meant to act as treatments. Think of them as tiny medical packages. They carry specific healing instructions directly to injured lungs. This approach flips the problem around. Instead of just blocking bad signals, we send good ones.
The process starts with a source of healthy cells. Researchers often use stem cells. These cells naturally release many exosomes. Scientists grow these cells in special containers. They collect the exosomes the cells shed. This gives a pure batch of empty delivery vehicles.
Next, researchers load these vesicles with helpful cargo. The cargo is chosen to fight ARDS. Common loads include: – Anti-inflammatory molecules to calm the immune system. – Growth factors to help repair damaged lung tissue. – Special RNA that can turn off harmful genes in target cells.
Loading can happen in two main ways. One method loads the parent cell. Scientists put the therapeutic cargo into the stem cell first. The cell then packages it into exosomes as they form. The other method loads exosomes after they are collected. Scientists use electrical pulses or gentle mixing. This opens temporary holes in the vesicle walls. The medicine slips inside.
The final step is targeting. An exosome must find the right address. Blanket delivery is inefficient. Researchers attach homing signals to the exosome’s surface. These are often small protein pieces called peptides. These peptides bind only to markers found on inflamed lung cells or blood vessels.
In ARDS, these engineered vesicles have clear jobs. They can deliver their calming cargo directly to immune cells gone wild. This tells macrophages to stop attacking. They can also bring repair tools to alveolar epithelial cells. These are the lung’s lining cells that get damaged.
Using shed exosomes this way is smart. The body already uses them for transport. Engineered versions blend in. They are less likely to be attacked by the patient’s immune system than synthetic nanoparticles. Their natural membranes help them fuse with target cells. This ensures the payload gets delivered inside.
Early tests in animal models show promise. Treated animals often have less lung fluid. Their oxygen levels improve faster. Inflammation markers in their blood drop. The engineered exosomes seem to follow their instructions.
This strategy faces hurdles too. Manufacturing enough for human patients is complex. Controlling the exact dose of medicine in each vesicle is hard. We must ensure these packages do not go to the wrong organ.
Still, the vision is powerful. It turns a part of the disease process into a cure. The same communication system that spreads injury could spread healing. Future work will refine these biological packages. The goal is to make them precise, safe, and strong enough for clinical use.
The next question is about timing and delivery. How would such a treatment actually reach a patient in need?
Delivering Anti-Inflammatory Agents via Exosomes
Exosomes can carry powerful anti-inflammatory medicines directly to injured lungs. These tiny vesicles act as natural delivery trucks. Their cargo can calm a dangerous immune overreaction.
Scientists load these shed exosomes with specific healing agents. These agents are not new drugs. They are often molecules our bodies already make. The exosome simply delivers them to the right place at the right time.
One key cargo is anti-inflammatory proteins. For example, exosomes can carry interleukin-10. This is a strong signal that tells immune cells to stop attacking. They can also deliver enzyme inhibitors. These blockers halt the production of harmful molecules that cause swelling and leaky blood vessels.
Another major cargo is nucleic acids. This includes microRNAs. These are small pieces of genetic code. They do not carry instructions for building proteins. Instead, they act like master switches. They can turn off genes that fuel inflammation.
Loading this cargo requires clever methods. Scientists often use electroporation. This technique uses a quick electric pulse to open tiny holes in the exosome’s membrane. The therapeutic molecules then slip inside. The holes quickly seal shut. Other methods incubate exosomes with high concentrations of the desired cargo. The molecules naturally pass through the membrane.
The beauty of this system is targeting. Exosomes from certain cells have a natural homing ability. Mesenchymal stem cell exosomes often travel to inflamed tissue. Scientists can also engineer the exosome surface. They can add small protein tags. These tags act like address labels. They guide the vesicle to lung cells.
Once delivered, the cargo gets to work inside the target cell. An anti-inflammatory protein can immediately start blocking signals. A microRNA can quickly silence a problematic gene. This action happens inside the cell where it is most effective.
The strategy has clear advantages over traditional drugs. – It protects fragile cargo. RNA molecules are easily destroyed in the bloodstream. The exosome’s lipid bilayer shields them. – It reduces side effects. Medicine is concentrated at the injury site. Less of it circulates to other organs. – It works with biology. The body’s own transport system is used for repair.
Research shows this approach can change key outcomes in models of lung injury. Delivering anti-inflammatory microRNAs via exosomes has been shown to reduce neutrophil infiltration. These are aggressive white blood cells that damage lung tissue. Studies also report lower levels of inflammatory cytokines in lung fluid.
The process from loading to effect involves several steps. First, exosomes are collected from cell cultures. Next, they are purified and loaded with the therapeutic agent. Then, they are administered, often through an intravenous injection. Finally, they navigate to the lung and release their cargo into target cells.
This method turns exosomes into precision tools. They move beyond simple carriers. They become active mediators of healing. Their natural composition helps them avoid immune detection. This allows for repeated dosing if needed.
The next challenge is scaling this precise system for human treatment. Manufacturing must ensure every batch carries a consistent, potent dose of medicine for reliable patient care.
Using Exosomes to Promote Lung Tissue Repair
Healing a damaged lung requires more than calming inflammation. The delicate architecture of the air sacs, the alveoli, must be physically rebuilt. This is where shed exosomes show immense promise. They carry direct instructions for repair.
After injury, cells release special exosomes. These vesicles are packed with signals. The signals tell the body to start fixing itself. They target two key cell types for lung recovery.
First, they help regenerate the alveolar epithelium. This is the thin lining of the air sacs. Specific growth factors and proteins in exosomes stimulate surviving epithelial cells. These cells then begin to divide and cover exposed areas.
Second, exosomes aid endothelial repair. The endothelium lines blood vessels. Leaky vessels worsen ARDS fluid buildup. Repair signals from exosomes help restore a tight barrier. This reduces harmful fluid in the lung spaces.
The cargo inside these repair exosomes is crucial. It often includes: – Growth factors like VEGF and HGF. These promote new cell growth. – Lipids that help form new cell membranes. – MicroRNAs that turn on pro-repair genetic programs. – Structural proteins like collagen for tissue scaffolding.
This cargo works in a coordinated way. It does not just deliver one drug. It delivers an entire toolkit. The body’s cells know how to use this toolkit naturally.
Research in animal models provides clear examples. Scientists collect exosomes from stem cells. They then inject these vesicles into injured lungs. The results are measurable. Lung tissue shows thicker, healthier alveolar walls. Blood gas levels improve, meaning better oxygen exchange. Scarring, or fibrosis, is often reduced.
The process mirrors natural recovery but accelerates it. The body already uses shed exosomes ards for communication. Therapeutic strategies simply enhance this existing system. They provide a concentrated dose of repair signals exactly where needed.
A key advantage is timing. Anti-inflammatory exosomes might be given early to stop damage. Pro-repair exosomes could follow later to rebuild. This two-step approach treats both the cause and the consequence.
Manufacturing these repair exosomes presents unique needs. The parent cells must be in a state that promotes healing messages. The conditions of cell culture matter greatly. Scientists are learning to “educate” cells to produce the most potent repair vesicles.
The ultimate goal is to restore lung function fully. This means not just survival but a return to normal life. Using exosomes to promote tissue repair brings that goal closer. It shifts treatment from managing failure to enabling true regeneration.
The next logical step is combining these approaches into a unified treatment protocol.
Future Directions and Clinical Implications
Current Research on Exosome-Based Diagnostics
Doctors need better tools to spot ARDS early. The condition often develops quickly in patients already very sick. Current methods rely on chest imaging and blood oxygen levels. These signs appear only after significant lung damage has already occurred. Researchers are now turning to exosomes for a solution. These tiny messengers travel through the bloodstream. They carry molecular cargo from their parent cells, including from injured lungs. This makes them perfect liquid biopsies.
Scientists are analyzing exosomes shed during the initial inflammatory phase of ARDS. They look for specific biological markers, or biomarkers, inside these vesicles. The goal is to find a clear molecular signature. This signature would act as an early warning signal. It could appear before traditional symptoms are obvious. Teams isolate exosomes from simple blood samples taken from at-risk patients. They then examine the contents with advanced tools.
The research focuses on several key components within these shed exosomes: – Unique microRNAs that control inflammation. – Proteins from damaged lung tissue. – Surface markers indicating which cell released the vesicle.
Finding the right combination is crucial. A single biomarker is rarely enough. The pattern matters most. For instance, a high level of one microRNA mixed with a specific protein could signal that alveolar cells are under severe stress. Another pattern might show that immune cells are becoming overactive. These profiles help doctors understand what is happening deep inside the lungs.
Recent studies show promising results. One project identified a panel of three microRNAs common in ARDS patients. These were not found in similar levels in people with other lung conditions. Another study tracked protein changes in exosomes over time. The shifts in protein content correlated with whether a patient’s condition improved or worsened. This suggests exosomes could be used for monitoring, not just diagnosis.
The practical benefits are significant. A reliable exosome-based test would be fast and minimally invasive. It requires only a small blood draw. Results could potentially come back in hours. This speed allows for quicker treatment decisions. Early intervention is critical in ARDS. Starting supportive care sooner can dramatically improve outcomes.
Challenges remain before this becomes routine. Researchers must validate these biomarkers in larger, diverse patient groups. They need to ensure the tests are consistent and affordable. The technology for rapid exosome analysis is still being refined for hospital labs. However, the path forward is clear. Exosome diagnostics represent a powerful shift toward precision medicine in critical care.
This diagnostic capability naturally complements therapeutic approaches. Identifying patients early creates a window for targeted interventions. The same vesicles that carry disease signals might one day be used to deliver treatment. Understanding what exosomes reveal about disease stage helps doctors choose the right therapy at the right time. This seamless link between diagnosis and action is the future of managing complex syndromes like ARDS.
Therapeutic Exosomes in Preclinical and Clinical Trials
The same shed exosomes that carry disease signals in ARDS are now being engineered as potential treatments. Scientists are actively redesigning these natural vesicles. The goal is to turn a problem into a solution.
Researchers can load exosomes with helpful cargo. This cargo aims to calm the overactive immune response in damaged lungs. Think of them as tiny, smart delivery trucks. They are programmed to go to the site of injury.
What can these therapeutic exosomes carry? The list is promising. – Anti-inflammatory molecules to reduce swelling. – Healing growth factors to help repair lung tissue. – Special RNA molecules that can silence harmful genes.
Preclinical studies in animal models show significant promise. In mice with lung injury, engineered exosomes have reduced fluid buildup. They have lowered levels of damaging cytokines. These are the proteins that drive the uncontrolled inflammation. Treated animals often show better oxygen levels and survival rates.
The journey from lab to clinic involves strict steps. First comes preclinical testing in animals. If results are safe and effective, researchers plan human trials. Early-phase clinical trials focus primarily on safety. Doctors give a small dose to a small group of volunteers. They watch for any side effects.
Later-phase trials test for effectiveness and ideal dosage. Several early-stage trials are now recruiting patients. These trials often involve exosomes derived from specific cell types, like stem cells. These natural exosomes may have inherent healing properties without extra engineering.
A key advantage is targeting. Exosomes naturally travel to inflamed tissues. This targeted delivery means the therapy could work where it is needed most. It might also require a lower dose than a standard drug circulating everywhere.
Challenges are significant for these new therapies. Manufacturing must be consistent and scalable. Regulators must agree on quality standards for these biological products. The long-term effects also need careful study.
The potential is a two-pronged approach. Doctors could one day use shed exosomes for diagnosis and monitoring. Then they could use engineered therapeutic exosomes for treatment. This creates a closed loop of precision care.
The road ahead is long but mapped. Preclinical data provides a strong foundation for hope. Ongoing clinical trials will deliver the crucial answers. Their results will determine if this elegant scientific idea becomes a routine life-saving tool.
Challenges in Exosome Therapy Development
Turning exosome science into reliable medicine faces major technical challenges. Scientists must solve these problems before treatments become widely available. The first big hurdle is making exosomes consistently. Cells naturally shed exosomes in small amounts. Producing enough for millions of patients is difficult.
Manufacturing must be both large-scale and pure. Imagine trying to collect tiny, identical bubbles from a giant vat of living cells. The process needs to be perfectly controlled every time. Even small changes can alter the exosomes. This affects their safety and how well they work.
Another challenge is isolation and characterization. Exosomes are extremely small. They must be separated from other cell debris and proteins. Scientists use complex methods like ultracentrifugation for this task. These methods are slow and not ideal for large batches. Newer techniques are being developed. All isolated exosomes must then be thoroughly checked. Researchers need to confirm their size, what cargo they carry, and their surface markers. This quality control is essential but adds time and cost.
Storage and delivery present further obstacles. Exosomes are fragile biological structures. Finding ways to freeze them without damage is critical. They must remain stable during transport from a factory to a hospital. Once in the body, targeting is another issue. While exosomes naturally go to inflamed areas, this homing is not perfect. Ensuring they reach the right lung cells in ARDS requires smart design.
Scientists are exploring ways to improve targeting. One method is engineering the exosome’s surface. They can add special guiding molecules to the membrane. These molecules act like address labels. They direct the exosome to specific damaged tissues.
The body’s immune system adds another layer of complexity. Could repeated doses cause an immune reaction? Researchers must study this risk carefully. They need to understand how long exosomes last in the bloodstream too. Their therapeutic effect depends on their survival time.
Regulatory pathways are also new and unclear. Exosomes are not simple chemical drugs. They are complex biological vesicles. Agencies like the FDA are creating new guidelines for them. Companies must prove their manufacturing is consistent. They must show their product is pure and potent in every batch.
Cost is a significant practical barrier. The current technology for making clinical-grade exosomes is expensive. This could limit patient access if not improved. Solving these problems requires collaboration across many fields. Biologists, engineers, and clinicians must work together.
Progress is being made on all these fronts despite the difficulties. Each solved challenge brings us closer to a viable therapy. The next phase will examine how overcoming these hurdles could change patient care fundamentally.
Personalized Medicine Approaches Using Exosome Profiles
Every patient’s battle with ARDS is unique. Their exosomes tell that personal story. These tiny vesicles carry specific molecular messages from their cells. Doctors could one day read these messages. This reading would guide precise, personalized treatment.
Think of it as a molecular fingerprint. The exosomes shed by a patient’s lung cells during ARDS are not random. Their cargo changes based on the stage and cause of inflammation. Some exosomes might carry signals that make inflammation worse. Others might carry signals that help repair tissue. The balance between these signals is key.
A blood test could analyze this exosome profile. It would look for specific biomarkers inside the vesicles. Biomarkers are biological red flags. Finding them early is crucial.
- A profile high in certain inflammatory proteins might show a patient needs strong anti-inflammatory drugs.
- A profile rich in repair factors might suggest the body is already healing. Treatment could then be less aggressive.
- A profile showing signs of bacterial infection could direct antibiotic choice.
This approach moves medicine from reactive to proactive. Instead of waiting for a patient’s oxygen levels to drop, doctors could adjust therapy based on the exosome forecast. Treatment becomes dynamic, not static.
Personalized medicine also applies to exosome therapies themselves. Not all therapeutic exosomes would be the same for every person. They could be loaded with cargo matched to a patient’s specific needs.
For example, one patient might need exosomes filled with molecules to calm an immune overreaction. Another might need exosomes carrying instructions to rebuild damaged blood vessels in the lungs. The source of the therapeutic exosomes could also be tailored. A patient’s own cells might be used to grow personalized vesicles. This could reduce immune rejection risks mentioned earlier.
The path to this future requires robust diagnostic tools. Researchers must first identify which exosome biomarkers are most reliable. They need to link specific profiles to clear clinical outcomes. Large studies will map these patterns.
Cost and speed are practical hurdles. Profiling exosomes must become fast and affordable for hospital labs. Advances in microfluidic chips and AI analysis are making this possible. These tools can sort and analyze exosomes quickly.
Ultimately, this shifts the treatment paradigm. ARDS management becomes a tailored strategy. Doctors use real-time biological data from shed exosomes to make decisions. This promises better outcomes with fewer side effects. It turns exosomes from just a potential therapy into an essential guide for all care. The next step is integrating this diagnostic power into clinical trials and everyday practice.
Practical Takeaways for Patients and Families
What the Exosome Research Means for ARDS Outcomes
Research into shed exosomes is changing what a severe ARDS diagnosis can mean. It offers new paths to better outcomes. This science moves beyond general support. It aims for precise treatment.
Think of exosomes as tiny messengers in a patient’s blood. These messengers come from inflamed lungs. They carry specific signals. Doctors can now read these signals. This gives them a real-time report on the disease’s activity. It is like getting live updates from the battle inside the lungs.
This information leads to practical changes in care. Here is how.
First, treatment can become more targeted. Currently, ARDS treatment is often similar for all patients. It supports the body while the lungs heal. Exosome analysis could change this. Doctors might see which harmful process is most active in a specific patient. One patient’s exosomes might show a raging immune attack. Another’s might reveal signals causing blood vessels to leak.
Therapy could then address these specific issues. This targeted approach may help stop damage faster. It could shorten the time a patient needs a ventilator.
Second, this science helps predict recovery. The mix of molecules in shed exosomes can serve as a forecast. Certain profiles may show that the body is starting to repair itself. Other profiles might warn of coming complications. This gives families and doctors a clearer picture. They can prepare for the next steps in the hospital stay or rehab.
For families, this means less guessing. It means more informed conversations with the medical team.
Third, it opens doors to new kinds of therapies. We are not just talking about diagnosis. The exosomes themselves could be used as treatment. Scientists are exploring how to design therapeutic exosomes. These could be given to a patient to fix problems.
For example, they could carry instructions to reduce scarring. They might tell cells to seal damaged blood vessels. This is not standard care today. But it is a direct result of understanding how natural exosomes work.
What does this mean for survival and long-term health? The goals are clear: – Reduce mortality by catching complications earlier. – Decrease the number of days spent in the intensive care unit. – Limit permanent lung damage after recovery. – Improve the quality of life for survivors.
This research brings a new sense of hope. ARDS is a complex syndrome. It varies greatly from person to person. Exosome science respects that complexity. It seeks to match the treatment to the individual’s unique biology.
The journey from lab to bedside continues. Yet the direction is promising. Future ARDS care will likely involve routine blood tests for exosome signals. These tests will guide daily decisions. They will help tell if a treatment is working or if a new approach is needed.
This turns a passive wait into an active strategy. For patients and families, that active strategy is the key to better outcomes. It represents a move toward precision medicine in one of critical care’s toughest challenges. The next phase involves validating these tools in large patient groups to make them standard practice.
How New Diagnostics Could Change Emergency Care
Imagine an ambulance rushing a patient with severe pneumonia to the hospital. Today, doctors face a critical delay. They must wait for ARDS to fully develop before confirming the diagnosis. This can take hours or even a day. New diagnostics aim to change that timeline completely.
The key is in the blood. Inflamed lungs shed exosomes into the bloodstream almost immediately. These tiny vesicles act as urgent messengers. They carry molecular signals from the site of injury. A rapid blood test could detect these specific exosomes right in the emergency room.
This changes everything. A positive test would signal that a dangerous immune overreaction has already begun. Doctors would not need to wait for worsening oxygen levels or a full chest X-ray picture. They could identify high-risk patients at the door.
Here is how emergency care could improve with this new tool.
- Faster treatment decisions: Therapies could start hours earlier. Early action is crucial in ARDS.
- Better patient sorting: Not everyone with pneumonia gets ARDS. A test could show who needs intensive care immediately.
- Reduced guesswork: Treatment becomes targeted, not general.
Consider the current standard. Doctors rely on oxygen levels and imaging. These signs appear only after significant lung damage has occurred. It is like seeing smoke after a fire has grown. Exosome signals are more like a heat sensor. They detect the spark.
The test itself might work like a common lab panel. A small blood sample is analyzed for specific exosome markers. A result could be ready within an hour. This speed turns diagnostic information into a real-time tool.
For families, this means less waiting in uncertainty. A clear result helps doctors explain the situation quickly. It provides a biological reason for why a patient is moving to the ICU. Understanding can reduce fear.
For patients, earlier intervention could prevent the worst damage. The goal is to stop the inflammatory cascade before it spirals out of control. This is the core promise of precision medicine in emergencies.
Hospitals would need new protocols. Emergency teams would learn to use this new data point. They would integrate it with other vital signs. This integration creates a sharper clinical picture.
The path from severe infection to organ failure might be intercepted sooner. This interception is the main practical takeaway. It shifts care from reactive to proactive at the most critical time.
The technology is still in development. But its potential to reshape emergency pathways is clear. It represents a fundamental shift in how we detect biological danger. The next challenge is making these tests robust and affordable for every hospital.
The Promise of Safer, Targeted Therapies Ahead
The real power of understanding shed exosomes in ARDS lies in future treatments. Current care for severe lung inflammation is mostly supportive. Doctors use ventilators and manage fluids. These methods treat the symptoms. They do not stop the underlying biological storm. Exosome research aims to change that.
Think of these tiny vesicles as messages. In ARDS, harmful messages spread fire through the lungs. But scientists are learning to send new instructions. They can engineer therapeutic exosomes in a lab. These designed vesicles could carry specific orders to cells.
The goal is to make treatments far safer and more targeted. Today’s strong anti-inflammatory drugs affect the whole body. This can cause major side effects. Targeted exosome therapy would work differently. It would deliver its cargo directly to injured lung tissue.
How could this work in practice? One approach uses exosomes as decoys. These decoys would bind to harmful molecules. They would stop these molecules from reaching and damaging lung cells. Another method uses exosomes as repair kits. They could deliver healing signals directly to the cells that line the air sacs.
- They might tell cells to strengthen their barriers.
- They could instruct immune cells to calm down.
- They may deliver materials to help rebuild damaged tissue.
This precision reduces collateral damage. It focuses the therapy where it is needed most. The body’s own communication system becomes the delivery truck.
Research is exploring specific cargo for these vesicles. Some carry molecules that silence harmful genes. Others bring proteins that promote tissue healing. The variety is vast. Each cargo type addresses a different part of the ARDS puzzle.
The path from lab to bedside has steps. First, scientists must identify the best source for these therapeutic exosomes. Next, they learn how to load them with the right instructions. Then, they must ensure the vesicles travel to the lungs after an intravenous injection.
Safety is a primary concern. Using natural biological vehicles lowers risk. The body already produces trillions of exosomes daily. Therapeutic versions would mimic this natural process. Their effect would be potent but localized.
For families, this future means hope beyond life support. It shifts the narrative from just surviving to active healing. A therapy that targets the root cause could shorten ICU stays. It could prevent long-term lung scarring known as fibrosis.
The timeline for such treatments is still years away. Current work focuses on robust laboratory studies and careful animal research. The promise, however, is a new class of smart medicines. These medicines would intercept disease with cellular precision.
This vision completes a powerful cycle. Advanced detection finds the biological spark early. Targeted therapy then works to extinguish it precisely. Together, they represent a new frontier in critical care medicine. The next challenge is turning this precise biological insight into a reliable, standard treatment for every emergency room.
Supporting Research and Staying Informed
Scientific progress moves quickly. Families can feel left behind. Staying informed is a powerful way to regain a sense of agency. Knowledge helps you ask better questions. It helps you understand treatment plans. You can also support the broader research mission.
Start with trusted sources. Look for major medical institutions. Universities and teaching hospitals often publish patient-friendly research summaries. Government health agencies are another excellent resource. These sites explain complex topics in clear language. They avoid hype and stick to proven facts.
Learn the key terms. Knowing basic vocabulary helps you search effectively. “Acute Respiratory Distress Syndrome” is the full name of ARDS. “Biomarkers” are biological warning signs. “Exosomes” are tiny message-carrying bubbles. Cells shed exosomes constantly. In ARDS, these vesicles carry signals that can worsen inflammation. Understanding this process is a big step.
Follow updates from research foundations. Non-profit groups dedicated to lung disease or critical care fund new studies. They share breakthroughs in newsletters and blogs. These updates are often more current than general news articles. They focus on the science, not just the headlines.
When reading about new discoveries, look for specifics. Good science reporting will mention the study phase. Was it in lab cells, animals, or humans? It will note the journal name. Reputable journals mean the work was reviewed by other experts. Be cautious of claims that sound too good to be true. Real progress is usually measured in small, careful steps.
You can also participate directly. Consider joining a patient registry. Registries collect health data from many people. This data helps scientists spot patterns. It can speed up future clinical trials. Ask your care team if they know of any ARDS registries or studies.
Discuss research with doctors respectfully. You might say, “I read about new work on shed exosomes in ARDS. Can you help me understand what this might mean for my loved one’s care?” This opens a dialogue. It shows you are engaged. Your doctor can provide crucial context for your specific situation.
Supporting research does not require a science degree. It requires curiosity and critical thinking. Your informed perspective is valuable. It contributes to a community that pushes for better treatments. This active role turns waiting into purposeful engagement.
The journey from lab to patient needs public understanding and support. Informed families help create demand for advanced therapies. They become partners in the long road toward new cures. This partnership is essential for turning today’s precise biological insight into tomorrow’s standard care.